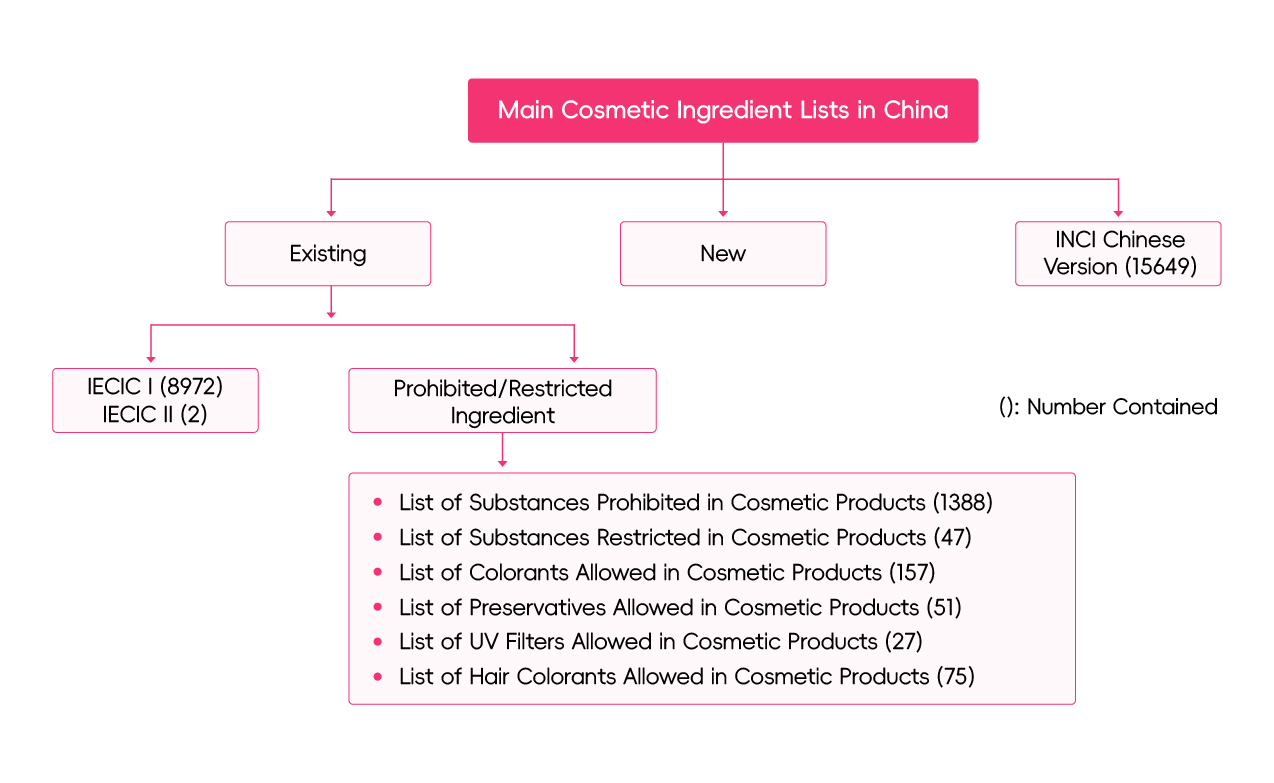
In China, cosmetic ingredients are divided into two categories: new cosmetic ingredients and existing cosmetic ingredients. Inventory of Existing Cosmetic Ingredients in China (IECIC) is an objective and comprehensive collection of existing ingredients used in cosmetics produced and sold in China. It serves as the sole reference of the regulatory status of cosmetic ingredients. All ingredients excluded from the Inventory are deemed to "new ingredients" and subject to mandatory pre-market registration or notification with the NMPA.
Part I The History of IECIC
According to Chinese cosmetic regulations, new cosmetic ingredients to be used in the products are required to complete pre-market approval, hence cosmetic manufacturers need to judge whether an ingredient is new prior to manufacturing. Before 2003, cosmetic ingredients were regulated based on the Cosmetics Hygienic Standards issued in 1987, which covered the regulated ingredients with prohibited substances, restricted substances, preservatives, UV Filters and colorants. Under this regulation, figuring out the status of an ingredient was difficult until the first publication of IECIC on 27 April 2003 by the Ministry of Health (MOH), the competent authority in charge of cosmetics at the time. Cosmetic ingredients listed in IECIC are regarded as existing ingredients and exempted from pre-market approval.
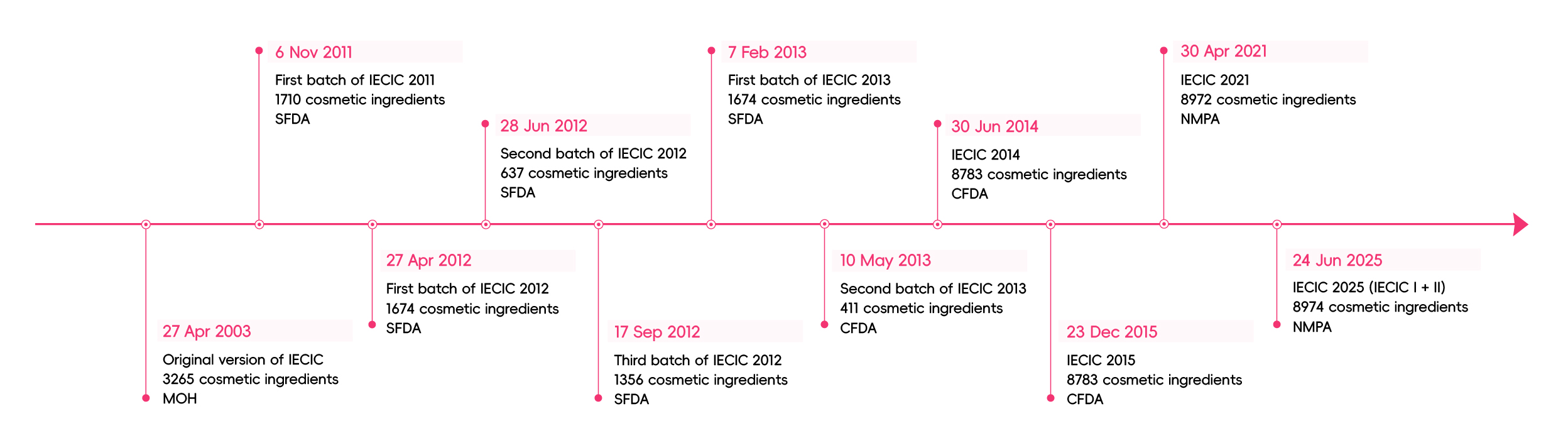 IECIC 2003 covered 3265 cosmetic ingredients that were used in China but was far from a comprehensive reflection of all ingredients used in the market. Besides, this Inventory did not align with international markets. From 1 Sep 2008, China Food and Drug Administration (CFDA) took over the full responsibility of cosmetic supervision from MOH. In order to guarantee safe use of existing ingredients and facilitate the determination and registration of new ingredients, CFDA started to amend IECIC.
IECIC 2003 covered 3265 cosmetic ingredients that were used in China but was far from a comprehensive reflection of all ingredients used in the market. Besides, this Inventory did not align with international markets. From 1 Sep 2008, China Food and Drug Administration (CFDA) took over the full responsibility of cosmetic supervision from MOH. In order to guarantee safe use of existing ingredients and facilitate the determination and registration of new ingredients, CFDA started to amend IECIC.
On 6 November 2011, CFDA issued the draft of IECIC 2011 for public consultation, which contained the first batch of existing ingredients in the Chinese cosmetic market, 1,710 in total. Compared with IECIC 2003, IECIC 2011 (first batch) only contained about a little more than half of the ingredients.
In 2012, CFDA released three draft batches of IECIC, totaling 3,667 ingredients. It was noted that the new inventory not only listed the Chinese names and the recorded names in International Nomenclature of Cosmetic Ingredients (INCI) of existing cosmetic ingredients, but also specified the limit for ingredients restricted to be used in cosmetics.
In 2013, CFDA finalized two batches of IECIC containing 2,085 substances in total. It was expected the third, which was also the last batch of IECIC 2013 will be published in the near future. However, CFDA chose to abandon the batch-wise release approach.
Instead of a batch-wise approach, CFDA decided to publish an integrated existing cosmetics inventory. On 30 June 2014, IECIC 2014 was released by CFDA, which was a consolidated list containing 8,783 existing cosmetic ingredients. This version is a significant enhancement in the number of existing cosmetic ingredients in comparison to the previous versions.
On 23 December 2015, CFDA released IECIC 2015, which added nine ingredients and deleted nine due to duplication errors. Therefore, the total number of ingredients in IECIC 2015 is the same as the IECIC 2014. The gradual improvement of IECIC has seen the use of cosmetic ingredients standardized, the determination and registration of new ingredients facilitated, and the safety of cosmetic products promoted.
On April 30, 2021, China’s National Medical Products Administration (NMPA, formerly CFDA) finalized the IECIC 2021 to align with the country’s newly implemented cosmetic regulatory framework, which took effect on January 1, 2021. The updated inventory officially entered into force on May 1, 2021, increasing the total number of existing cosmetic ingredients from 8,783 to 8,972.
Compared with its 2015 predecessor, IECIC 2021 not only included the Chinese name and INCI/English name of each ingredient, but also added data on their highest historical use concentration in rinse-off and leave-on products, as well as relevant notes. These concentration values served as recognized evidence for simplified safety assessments during the transitional phase of the implementation of China’s cosmetic safety assessment system.
Part II IECIC 2025 Reform
On June 24, 2025, the NMPA unveiled a significant reform of the IECIC, introducing a dual-list structure and a dynamic adjustment mechanism to further standardize cosmetic ingredient management.
1. Dual-List Structure
The IECIC is now divided into two separate lists: IECIC I and IECIC II.
IECIC I | It is a revised and improved version of the IECIC 2021, containing 8,972 ingredients that have been used in cosmetics manufactured or sold within China. Key updates include:
|
IECIC II | It is a newly established list for including new cosmetic ingredients (NCIs) which have completed their three-year safety monitoring period following registration or notification in China and now qualify for inclusion in the IECIC. The first two entries added to IECIC II are Acetylneuraminic Acid and Beta-Alanyl Hydroxyprolyldiaminobutyroyl Benzylamide. Compared with IECIC I, IECIC II provides more comprehensive technical data for each ingredient, including: Chinese and INCI/English Names, CAS Number, Molecular Formula, Structural Formula, Relative Molecular Mass, Intended Use, Safe Use Level. It is important to note that the "safe use level" provided in IECIC II is not equivalent to a legally mandated maximum concentration. A concentration exceeding this level this level does not necessarily indicate a product is unsafe, nor does staying within it guarantee safety. |
This structural reform aligns with Article 14 of the Cosmetic Supervision and Administration Regulation (CSAR), which permits the inclusion of NCIs in the IECIC after they have completed their safety monitoring period without any reported safety concerns. The dual-list structure offers a clear distinction between historically used ingredients and NCIs that have undergone scientific safety evaluation and monitoring, thereby providing more targeted and traceable ingredient management.
2. Dynamic Adjustment Mechanism
To improve the flexibility and efficiency of IECIC management, the NMPA has established a dynamic adjustment mechanism. Both IECIC I and IECIC II will now be continuously supplemented, revised, and corrected based on scientific research, industry development, and regulatory needs.
Moving forward, updates to the IECIC will no longer be announced via separate public notices. Instead, all revised versions and corresponding adjustment explanations will be published directly on the IECIC Query Page of the NMPA’s official website. This approach ensures timely, transparent, and convenient access to the most current ingredient information for both enterprises and the public.
Part III IECIC and the INCI Chinese Version
Cosmetic companies might confuse IECIC with the INCI Chinese Version and falsely regard ingredients in the latter as "used or existing" in China. It is worth clarifying that the INCI Chinese Version is translated to standardize the Chinese translation of INCI name, cosmetic labeling and instructions, which is certainly not a criterion for illustrating the regulatory status of an ingredient.
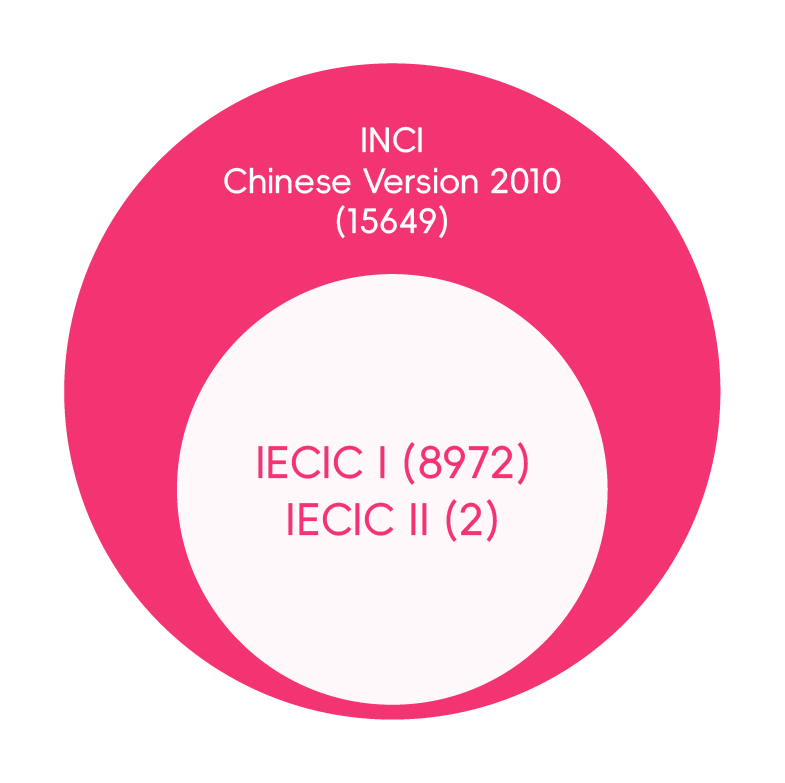 The INCI Chinese Version was first published by MOH in 2007 with 12,072 ingredients, which was translated according to the INCI names included in the International Cosmetic Ingredient Dictionary & Handbook Tenth Edition (2004) (ICI Dictionary) managed by the Personal Care Products Council (PCPC). The List is updated accordingly with the expansion of the ICI Dictionary.
The INCI Chinese Version was first published by MOH in 2007 with 12,072 ingredients, which was translated according to the INCI names included in the International Cosmetic Ingredient Dictionary & Handbook Tenth Edition (2004) (ICI Dictionary) managed by the Personal Care Products Council (PCPC). The List is updated accordingly with the expansion of the ICI Dictionary.
The latest INCI Chinese version containing 15,649 ingredients was translated and released by CFDA in 2010, based on its twelfth Edition (2008). Cosmetic manufacturers are required to label the ingredients using the standard Chinese names if they are covered by the List.
Now the NMPA is revising the 2010 version. It is expected that the new version will be released soon.




 Request a Demo
Request a Demo
