The promulgation of Cosmetic Supervision and Administration Regulation and its subsidiary rules, standards, and guidance documents in 2021 overhauled China’s cosmetic regulatory framework, bringing the industry into a new era of comprehensive and advanced supervision. In 2022, some transitional arrangements are about to expire, and some supporting regulations are about to take effect. To this end, ChemLinked sorted out the noteworthy regulatory matters and corresponding time points for cosmetic registration and notification in 2022 and beyond to help cosmetic stakeholders make relevant preparations in advance, so as to efficiently and orderly address compliance challenges and complete relevant notification and registration work.
Regulations Entering into Force in 2022
No. | Effective Date | Regulations | Brief Introduction |
1 | January 1, 2022 | Supervision and Administration Measures on Cosmetics Manufacture and Operation | The regulation clarifies the responsibilities of cosmetics registrants, notifiers, entrusted production enterprises, as well as the requirements of cosmetic manufacture management, operation management, etc. |
2 | Supervision and Administration Provisions on Children Cosmetics | The regulation specifies the scope, formula design principles, labelling requirements, registrant/notifier's main responsibilities, post-market supervision requirements, etc., of children cosmetics. | |
3 | May 1, 2022 | The regulation spells out the requirements for the labelling and prohibited claims of cosmetics (including cosmetics provided to consumers in the form of trial, gift, exchange, etc.) under China’s new cosmetic regulations. | |
4 | July 1, 2022 | The regulation specifies the requirements for all aspects of cosmetic production and quality control, such as ingredients, personnel, equipment, sanitation, inspection, packaging, storage, transportation, sales, and recall. |
Noteworthy Regulatory Matters and Corresponding Time Points 1
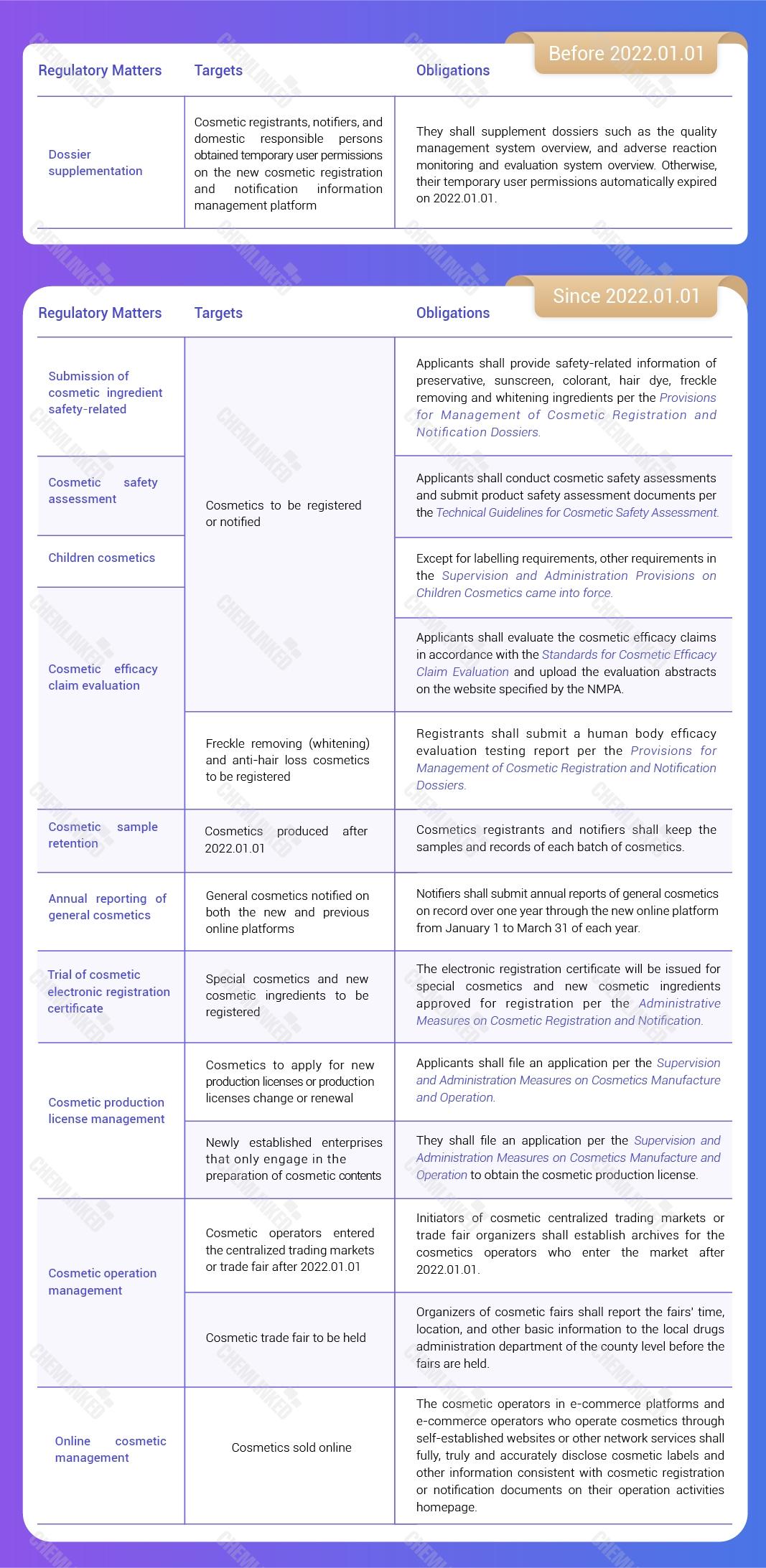
1. Temporary User Permissions on the New Platform
Cosmetic registrants, notifiers, and domestic responsible persons who have opened temporary user permissions on the new cosmetic registration and notification information management platform (“new platform”) should supplement dossiers such as the quality management system overview, and adverse reaction monitoring and evaluation system overview before January 1, 2022.
If they failed to supplement such dossiers within the time limit, their temporary user permissions would be automatically invalid on January 1, 2022. If relevant dossiers are completed in the future, they can still apply for opening the registration and notification user permissions.
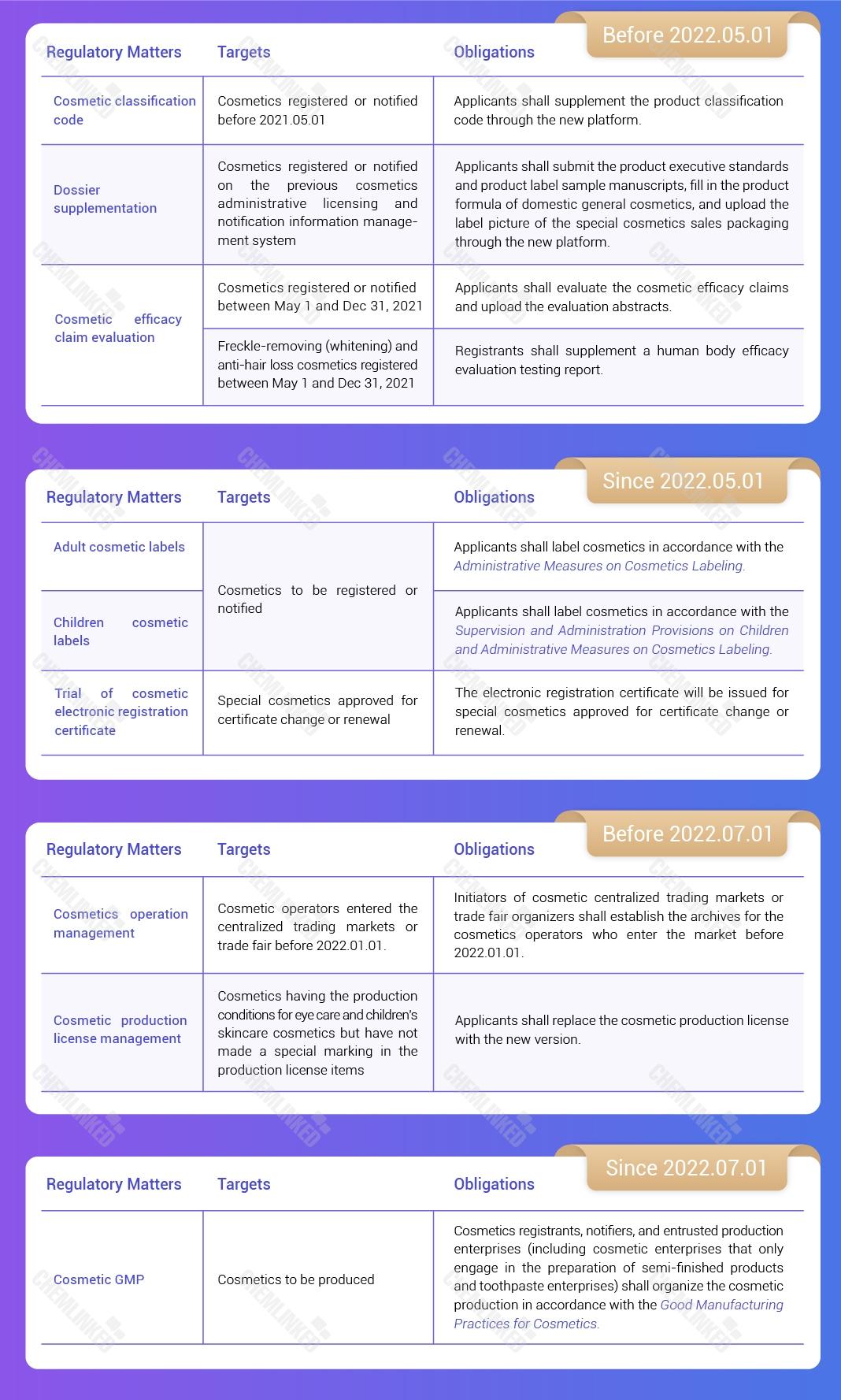
2. Dossier Supplementation of Products that Have been Registered and Notified on the Previous Platform
For cosmetics that have been registered or notified on the previous cosmetic administrative licensing and notification information management system (“previous platform”), the registrants and notifiers shall submit the product executive standards and product label sample manuscripts, fill in the product formula of domestic general cosmetics, and upload the label picture of the special cosmetics sales packaging through the new platform before May 1, 2022.
Product Classification | Dossiers Need to be Supplemented |
Imported General Cosmetics | The product classification code, product executive standards and product label sample manuscripts |
Domestic General Cosmetics | The product classification code, product executive standards, product label sample manuscripts, and the product formula with all ingredients’ content |
Special Cosmetics | The product classification code, product executive standards, product label sample manuscripts, and the label picture of the sales packaging |
Note: Given that the annual report of general cosmetics notified over one year shall be submitted before March 31, 2022, the dossier supplementation of general cosmetics shall be completed before March 31, 2022. | |
3. Cosmetic Classification Code
From May 1, 2021, the newly registered or notified cosmetics shall fill in the product classification code according to the Cosmetic Classification Rules and Catalogs.
For cosmetics that have been notified or registered before May 1, 2021, the registrant or notifier shall supplement the product classification code through the new platform before May 1, 2022.
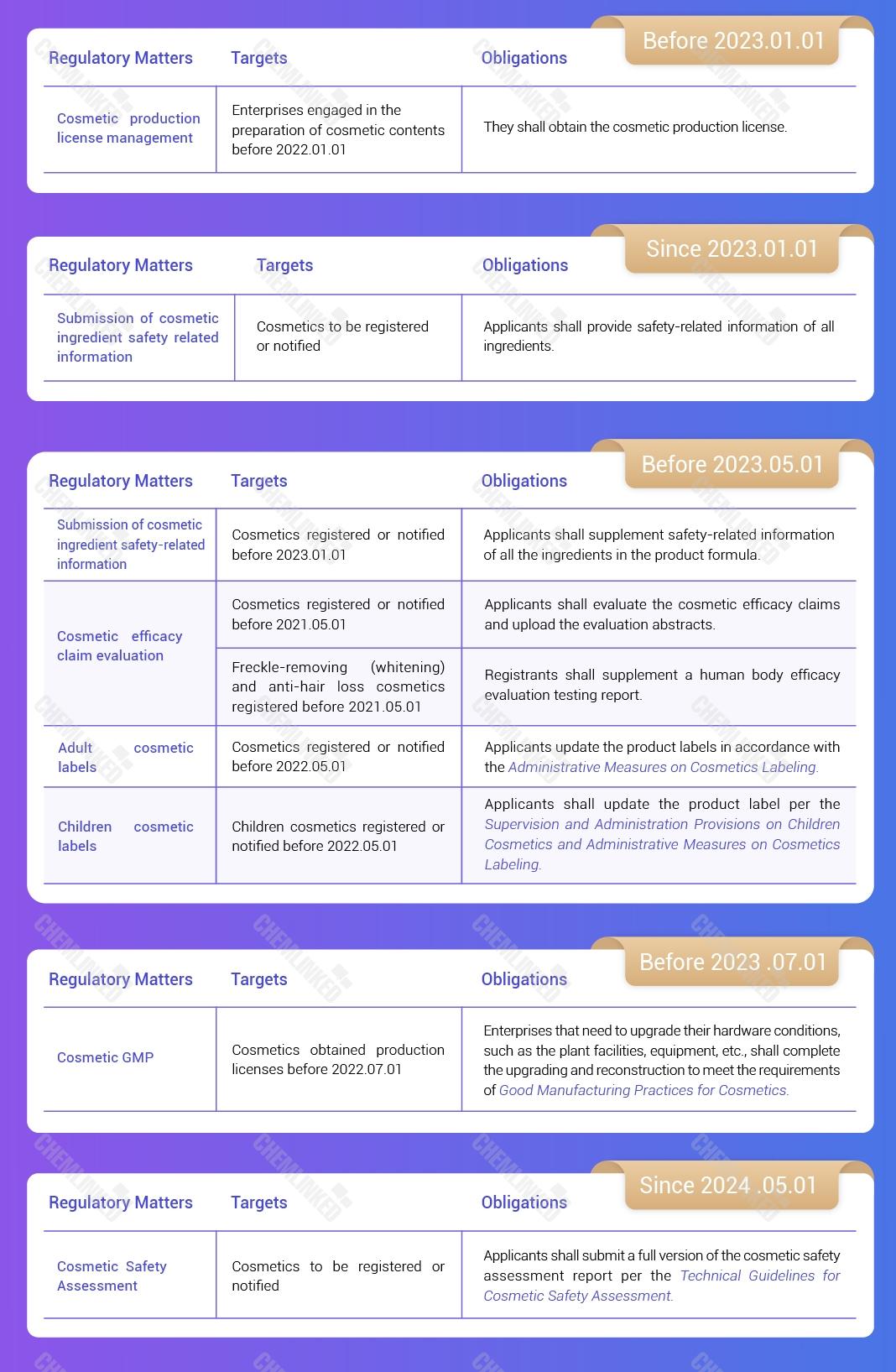
4. Submission of Cosmetic Ingredient Safety Related Information
Starting from January 1, 2022, the newly registered or notified cosmetics shall provide safety-related information of preservative, sunscreen, colorant, hair dye, freckle removing and whitening ingredients.
Starting from January 1, 2023, when applying for registration or notification, the registrants or the notifiers shall provide safety-related information of all ingredients.
For cosmetics that have been registered or notified before January 1, 2023, the registrant and notifier shall supplement safety-related information of all the ingredients in the product formula before May 1, 2023.
Notably, since 9 a.m. on December 31, 2021, the cosmetic ingredients safety information submission platform has been in service. Cosmetic ingredient manufacturers or their authorized enterprises can log on to the platform and submit ingredient safety-related information to obtain an ingredient submission code.
5. Annual Reporting of General Cosmetics
Starting from January 1, 2022, the annual report system has been implemented uniformly for general cosmetics notified through the previous and new platforms. Notifiers shall submit the annual report of general cosmetics notified over one year through the new platform from January 1 to March 31 of each year. Products that fail to complete the annual report in time will be forcibly cancelled.
The precondition for submitting the annual report is to open the user permission of registrant/notifier or domestic responsible person on the new system and complete the product confirmation. The annual report of general cosmetics shall include the following contents:
General situation of production and import of products, as well as the suspension of production during the period;
Self-inspection on the conformity of products with laws and regulations, mandatory national standards and technical specifications.
6. Cosmetic Safety Assessment
Starting from January 1, 2022, all newly registered or notified products shall conduct cosmetic safety assessments and submit product safety assessment documents per the requirements of the Technical Guidelines for Cosmetic Safety Assessment (“the Technical Guidelines”).
Before May 1, 2024, a simplified version of the cosmetic product safety assessment report can be submitted.
After May 1, 2024, all newly registered or notified products shall submit a full version of the cosmetic product safety assessment report in accordance with the requirements of the Technical Guidelines.
7. Cosmetic Efficacy Claim Evaluation
Starting from January 1, 2022, cosmetics registrants and notifiers shall evaluate the cosmetic efficacy claims in accordance with the Standards for Cosmetic Efficacy Claim Evaluation (“the Standards”) and upload the evaluation abstracts on the website specified by the NMPA.
For cosmetics registered or notified before May 1, 2021, the cosmetics registrants or notifiers shall evaluate the cosmetic efficacy claims in accordance with the Standards and upload the evaluation abstracts before May 1, 2023.
For cosmetics registered or notified between May 1, 2021 and December 31, 2021, the cosmetic registrants or notifiers shall evaluate the cosmetic efficacy claims in accordance with the Standards and upload the evaluation abstracts before May 1, 2022.
Regarding the Efficacy Evaluation Testing Report of Freckle-removing (Whitening) and Anti-hair Loss Cosmetics
Starting from January 1, 2022, when applying for the registration of freckle-removing (whitening) and anti-hair loss cosmetics, the registrants shall submit a human body efficacy evaluation testing report that meets the requirements of the Provisions for Management of Cosmetic Registration and Notification Dossiers.
For freckle-removing (whitening) and anti-hair loss cosmetics that have been registered before May 1, 2021, the registrants shall supplement a human body efficacy evaluation testing report before May 1, 2023.
For freckle-removing (whitening) and anti-hair loss cosmetics that have been registered between May 1, 2021 and December 31, 2021, the registrants shall supplement a human body efficacy evaluation testing report before May 1, 2022.
8. Cosmetic Labels
Starting from May 1, 2022, all cosmetics to be registered or notified shall be labelled in accordance with the Administrative Measures on Cosmetics Labeling (“the Measures”).
For cosmetics that have been registered or notified before May 1, 2022, the registrants or notifiers shall update the product labels in accordance with the Measure by May 1, 2023.
9. Children Cosmetics
Except for labelling requirements, other requirements in the Supervision and Administration Provisions on Children Cosmetics have come into force on January 1, 2022.
Children cosmetics that apply for registration or notification after May 1, 2022 must be labelled in accordance with the Supervision and Administration Provisions on Children Cosmetics and Administration Measures on Cosmetics Labelling.
For children cosmetics registered or notifier before May 1, 2022, the registrant or notifier shall update the product label before May 1, 2023.
10. Cosmetic Sample Retention
Cosmetics registrants and notifiers should keep the samples and records of each batch of cosmetic products produced after January 1, 2022. The reserved samples shall be kept in the previous sales packaging, and the quantity thereof shall meet the requirements of product quality inspection.
In the case of cosmetic entrusted production, the entrusted production enterprise shall also keep samples and records thereof per provisions of the Supervision and Administration Measures on Cosmetics Manufacture and Operation.
Overseas cosmetics registrants and notifiers shall keep the samples of each batch of products imported into China. The samples and records shall be kept by their responsible persons in China.
11. The Trial of Cosmetic Electronic Registration Certificate 2
From January 1, 2022, the electronic registration certificate has been issued for special cosmetics and new cosmetic ingredients approved for registration per the Administrative Measures on Cosmetic Registration and Notification.
From May 1, 2022, the electronic registration certificate will be issued for special cosmetics approved for certificate change or renewal.
Notably, the electronic registration certificate has the same legal effect as the paper registration certificate. During the trial period, the electronic and the paper registration certificate are issued in parallel.
12. Cosmetic Production License Management
Starting from January 1, 2022, the application for new cosmetic production licenses and the application for changing and renewing cosmetic production licenses shall be conducted per the Supervision and Administration Measures on Cosmetics Manufacture and Operation (“the Measures”).
The cosmetic production license obtained before January 1, 2022, will remain valid within the previous validity period, except for those that have the production conditions for eye care cosmetics and children's skincare and eye care cosmetics but have not specially marked the corresponding items on the licenses. These enterprises shall replace the cosmetic production license with the new version by July 1, 2022.
Starting from January 1, 2022, newly established enterprises that only engage in the preparation of cosmetic contents should file an application to the medical products administration departments of the provinces, autonomous regions, or municipalities directly under the Central Government where they are located. They can produce only after granted the cosmetic production license.
For enterprises engaged in the preparation of cosmetic contents before January 1, 2022, they shall obtain the cosmetic production license by January 1, 2023.
13. Cosmetic GMP
Starting from July 1, 2022, cosmetics registrants, notifiers, and entrusted production enterprises (including cosmetic enterprises that only engage in the preparation of semi-finished products and toothpaste enterprises) shall organize the cosmetic production in accordance with the Good Manufacturing Practices for Cosmetics (“the Practices”).
For enterprises that have obtained production licenses before July 1, 2022, and need to upgrade their hardware conditions such as the plant facilities, equipment, etc., the upgrading and reconstruction shall be completed according to the Practices before July 1, 2023.
14. Cosmetic Operation Management
Since January 1, 2022, initiators of cosmetic centralized trading markets and trade fair organizers shall establish archives for the cosmetics operators who enter the market after January 1, 2022.
For cosmetics operators who enter the market before January 1, 2022, the initiators or organizers shall establish the archives mentioned above by July 1, 2022.
Since January 1, 2022, the organizer of cosmetic fairs shall report the fairs' time, location, and other basic information to the local drugs administration department of the county level before the fairs are held.
15. Online Cosmetic Management
Starting from January 1, 2022, the cosmetic operators in e-commerce platforms and e-commerce operators who operate cosmetics through self-established websites or other network services shall fully, truly and accurately disclose cosmetic labels and other information consistent with cosmetic registration or notification documents on their operation activities homepage.
Tmall has issued a notice requiring merchants to display the front and back label pictures of cosmetic products sold on the product main image or product details page or SKU image, and fully and accurately fill in the cosmetic product attribute information such as the name and the address of the registrant or notifier, the name of the manufacturer, product executive standard number. The filled information shall be consistent with the registration or notification information.
Noteworthy Regulatory Matters in Chronological Order
ChemLinked Service
ChemLinked expects to assist companies in registering/notifying cosmetics, applying for ingredient submission codes, reviewing cosmetic labels, etc. in China. If you have any further cosmetic compliance consultation needs, please contact us at [email protected].




 Request a Demo
Request a Demo
 We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 









