Takehome:
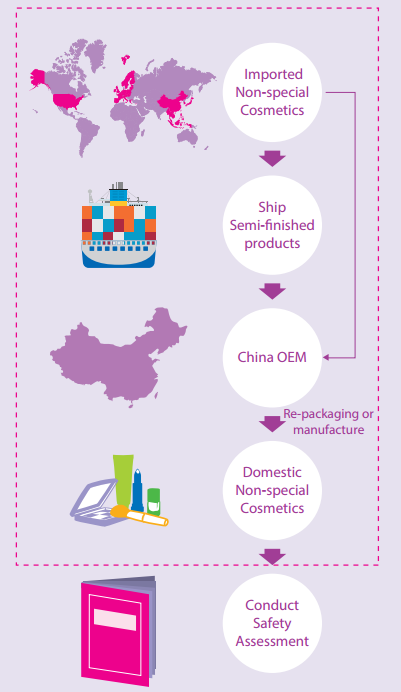
Our sources have reliably informed us that in the near future the sale of repackaged cosmetics will be prohibited by CFDA. In fact it has been revealed by some companies that filing of repacked cosmetics has already been rejected by Beijing FDA. Guangdong FDA has notified companies within its prefecture of the upcoming dramatic change but no specific enforcement rule or transitional measures have been issued. However, in some provinces e.g. Jiangsu, filing is still acceptable.
Changes to Regulatory Requirements
Industry first became aware of the changes in cosmetic regulatory requirements when companies applied for filing modification of repacked cosmetics. The notification released by the CFDA on Dec 15th 2015 stipulated that since January 1st 2016, Cosmetics manufacturers shall exchange National Industrial Manufacture License and Hygiene License for Cosmetics Manufacturers for one Cosmetics Manufacture License and complete the exchange before January 1st 2017. The new license holders are required to update existing packaging with the new license number necessitating; an application for filing modification. However some FDAs have outlined that repacking of cosmetics is banned after consolidation of the cosmetic manufacture license system. Beijing FDA has implemented a transitional measure under which the filing of cosmetics manufactured before the issuance date of a new license can be approved; otherwise the filing will be disapproved.
Enterprises that haven’t submitted the two licenses are hesitant to apply for the new license. If they don’t apply for it, they can still repack cosmetics since there is no need to update the packaging. However once a new license is granted their cosmetic filing application will likely fail under the pending requirements.
Industry Implications:
Since repackaging of cosmetics is widespread, the change will have a huge impact on the industry. The existing cosmetics that failed the filing cannot be marketed, which will cause large losses for OEMs and cosmetic brands. OEMs will have to cut their service of repacking and lose considerable profits. Cosmetic brands shall find another way to reduce costs and ensure the production process and quality control of their products. For the OEMs that obtained the new license and cosmetic brands that intend to continue or start subcontracting repacking, for now it may be advisable to suspend repacking projects and wait for clear provisions made by the CFDA.
Schedule Concerning Exchanging for Cosmetics Manufacture License
|
Time |
Obligation |
|
Since January 1st 2016 |
All new cosmetics manufacturers shall apply for a Cosmetics Manufacture License from provincial FDA |
|
Since January 1st 2016 |
Cosmetics manufacturers having obtained National Industrial Manufacture License from AQSIQ and Hygiene License for Cosmetics Manufacturers from provincial FDA can exchange their licenses for Cosmetics Manufacture License. |
|
Since July 1st 2017 |
Cosmetic products manufactured after the date must use new labels with information of Cosmetics Manufacture License. Current labels on packages can be used until June 30th 2017. |
It can be expected that by July 1st 2017 repacked cosmetics will disappear from the Chinese market provided the ban is implemented.
Circumventing China's Animal Testing Requirements: A Thing of the Past
In the past there were two ways to circumvent China's animal testing requirements: repack in China and cross-border e-commerce. However this April, China released the positive lists of CBEC Commodities and stipulated that cosmetics sold through CBEC shall obtain administrative license or filing certificate afterwards. This essentially means that CBEC will no longer be a way to circumvent China's animal testing and Chinese labels any more (see CL news on 15 Apr 2016).
If repackaging is banned another way of circumventing China’s animal testing requirements will disappear. The last way available will be to establish a factory in China or to fully subcontract production.




 Request a Demo
Request a Demo We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 







