BackgroundSince the implementation of the Cosmetic Supervision and Administration Regulation (CSAR) in 2021, the China National Medical Product Administration (NMPA) has introduced a risk-based classification system for cosmetic products and NCIs. High-risk NCIs must undergo registration with the NMPA, while low-risk NCIs only need to be notified and can subsequently be manufactured and imported once enterprises complete the online dossier submission. This streamlined process reduces the market entry threshold for NCIs in China.
However, challenges have arisen during the registration and notification process for NCIs. Some registrants and notifiers have encountered difficulties due to their incomplete understanding of the identification and categorization of NCIs as well as inadequate data preparation. Common mistakes include the unclear definitions of NCIs, NCIs’ functions exceeding the scope of cosmetic definitions, and the inaccurate determination of applicable situations for NCIs. |
To standardize and provide guidance for the determination and research of NCIs, the China National Institutes for Food and Drug Control (NIFDC) released the first draft of the Technical Guidelines for Determination of New Cosmetic Ingredients on January 19, 2023, to seek feedback from the public. 1
Taking industry input into account, NIFDC released the second draft, which was renamed to Technical Guidelines for Determination and Research of New Cosmetic Ingredients(Guidelines) on December 1, 2023. 2 Any suggestions can be submitted to [email protected] until December 18.
The Guidelines is divided into four parts: overview, categorization, general requirements, and explanation of terms. Notifiers and registrants should utilize the Guidelines to assess whether an ingredient belongs to NCI, determine the appropriate NCI category, and conduct research in alignment with the provided requirements.
NCI Categorization
The first draft primarily categorized NCIs based on four aspects: ingredient composition, source attribute, preparation process, and actual function. However, the second draft restructures the categorization, focusing on "management attributes," and "management categories." Additionally, the second draft provides clarity on "ingredients that are not considered NCIs."
1. Categorization based on Management Attributes
In terms of management attributes, the Guidelines categorizes NCIs into three groups: "NCIs that affect cosmetic products," "NCIs that affect the surface of the human body," and "NCIs that affect both products and the surface of the human body."
Category | Description | Research Requirements |
NCIs that affect cosmetic products | Ingredients with an effect on the dosage form, stability, color, odor, etc. of cosmetic products to enhance or keep its performance, and do not have impact on the human body within a certain range of action. This NCI category mainly includes matrix ingredients and auxiliary ingredients. | During the R&D process, it is crucial to analyze the performance of ingredients, with a primary focus on ensuring their safety and quality control. The appropriate usage amounts in cosmetic applications should be thoroughly studied. |
NCIs that affect the surface of the human body | Ingredients that have tangible effects on the skin, hair, nails, lips, and other surfaces of the human body, such as moisturizers, emollients, anti-wrinkle agents, whitening agents, sunscreens, hair dyes, anti-hair loss agents, etc., and can enable cosmetics to achieve purposes of cleansing, protection, beautification, and enhancement. | During the R&D process, it is essential to establish a clear understanding of the relationship between the intended purpose of the ingredient and its actual role in cosmetics. The primary focus is ensuring its safety, efficacy, and quality control. |
NCIs that affect both products and the surface of the human body | While exerting its effects on cosmetic products, this NCI category simultaneously affects the surface of the human body. | In addition to scientifically and accurately determining the intended purpose of these ingredients based on their characteristics and anticipated role in cosmetics, it is vital to prioritize effectiveness. Relevant research should be conducted to substantiate the ingredient's alignment with its designated purpose. |
2. Categorization based on Management Categories
The Guidelines categorizes NCIs into three management categories: "NCIs used for the first time in China and abroad," "NCIs used for the first time in China," and "NCIs based on improvements and innovations to existing ingredients." Each category of NCIs has distinct research requirements pertaining to preparation technique, stability testing, quality control standards, functional evaluation, and safety assessment.
Category | Description |
NCIs used for the first time in China and abroad | Ingredients that are newly discovered and confirmed to be suitable for being used in cosmetics, or ingredients that are known but have not yet been utilized in cosmetics in China and abroad. |
NCIs used for the first time in China | Ingredients that have been utilized in cosmetics internationally but have not yet been utilized in cosmetics in China. |
NCIs based on improvements and innovations to existing ingredients | 1) Existing ingredients with clearly defined purposes and safe usage amounts whose purposes are adjusted to preservatives, sunscreens, colorants, hair dyes, or freckle-removing and whitening agents; or 2) Existing ingredients with clearly defined purposes and safe usage amounts who are improved and innovated. To enhance safety, efficacy, stability, and quality control of the ingredients, companies can conduct substantial R&D on ingredients by optimizing ingredient preparation technology, refining production processes, and modifying the material foundations of the ingredients. |
Furthermore, the Guidelines provides details on the research content and naming requirements for NCIs in the "General Requirements" section. It also elucidates the necessary research to be conducted on different NCIs, including preparation technology, stability testing, quality control standards, functional evaluation, and safety assessment. For more detailed information on the research requirements, please feel free to contact us at [email protected] for translation service.
3. Ingredients that are not considered NCIs
The Guidelines outlines the criteria for identifying ingredients that are not considered NCIs.
1) Ingredients whose intended usage method, application site, or purpose in cosmetics exceeds the boundaries of the cosmetic’s definition;
2) Ingredients explicitly prohibited by cosmetic technical regulations; ingredients sharing similar basic structures, physical and chemical properties, and/or actual functions with prohibited ingredients; and ingredients deemed to be harmful to human health under normal, reasonable, and foreseeable conditions of use;
3) Ingredients listed in the Inventory of Existing Cosmetic Ingredients in China (IECIC), specific ingredients falling under group categories in the IECIC, and ingredients whose material foundation aligns with the ingredients listed in the IECIC.
Communication Mechanism for NCI Registration and Notification
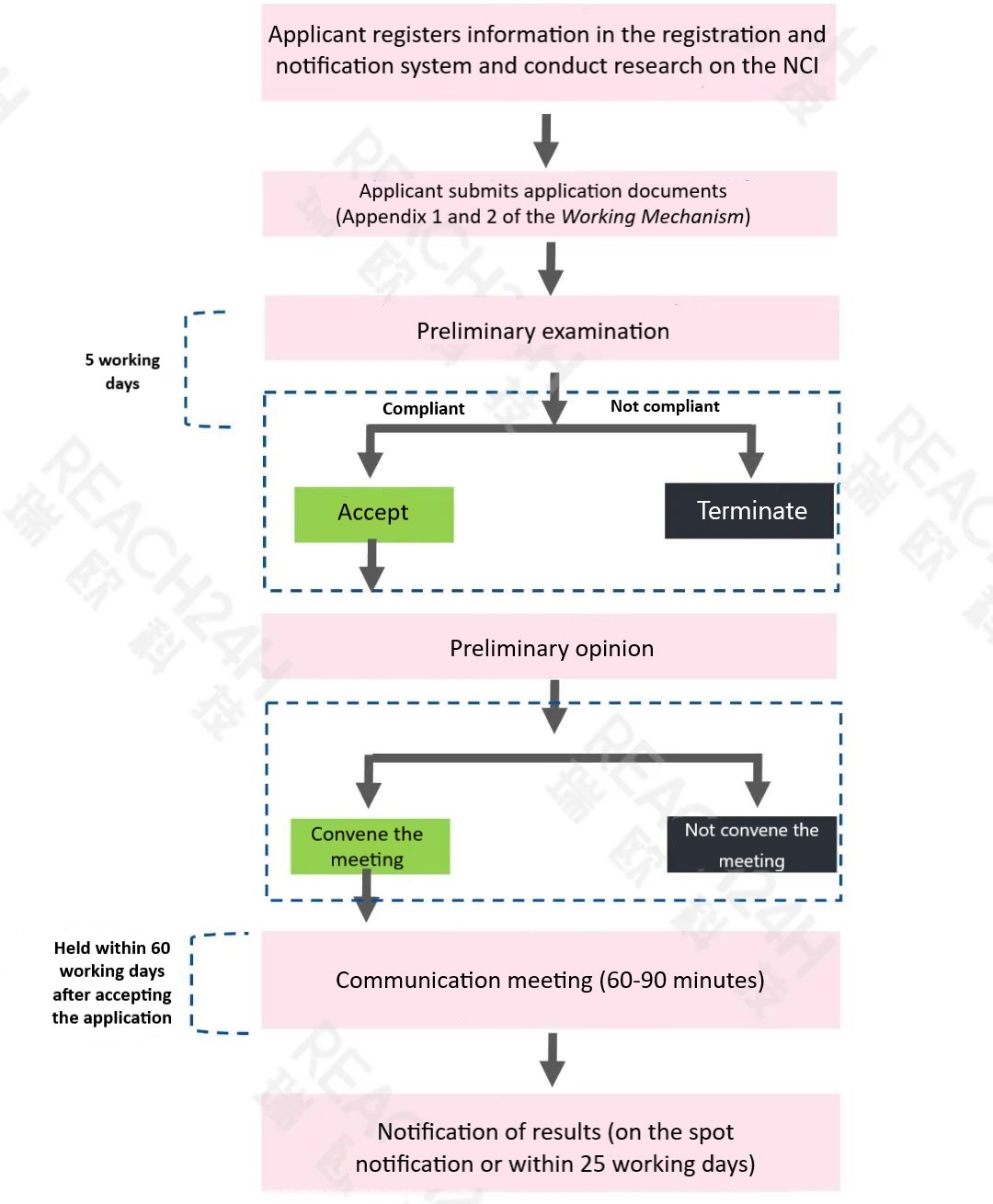
Concurrent with the release of the Guidelines, NIFDC introduced the Working Mechanism for Communication and Exchange of New Cosmetic Ingredients (Trial). 3 This mechanism aims to facilitate the communication between registrants, notifiers, and the review department regarding technical matters pertaining to NCIs prior to the submission of registration or notification applications, as well as during the review process. General inquiries can be made in person, via email, or over the phone, while crucial technical matters can be addressed through meetings, driving consensus with the review department.
Please refer to the following flow chart for the specific procedures.
Industry Comments
Industry insiders highlighted that prior to the release of the Guidelines, they had faced challenges in understanding the regulatory standards when applying for NCI notification. Even if an NCI had a history of overseas usage or domestic consumption, companies still adhered to the strictest standards and conducted comprehensive toxicological tests. They expect the implementation of the Guidelines and Communication Mechanism would reduce enterprise costs. 4
Ligang Jiang, the President of the Daily Chemicals Division at Zhejiang Huarui Biotechnology Co., Ltd., expressed that the registration and notification of NCIs had been a new thing in China, lacking experience both from the regulatory side and the enterprise side. 4 During the initial period, the NIFDC experts encountered some issues when reviewing application documents, such as the unclear definition of skin protectants, unclear definition of high activity, process authenticity, and effective dosage. Jiang stated, "Some companies' past submissions did not meet expectations, leading to further improvements in the regulations. The categorization of NCIs and separate treatment for different NCI categories can help clarify key review points."
Furthermore, according to Jiang, as regulations become increasingly stringent, it remains uncertain whether all notified NCIs will successfully pass the monitoring period. Therefore, companies planning to use notified NCIs are advised to select those with robust data to mitigate the risk of non-compliance with marketed products.




 Request a Demo
Request a Demo
 We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










