On October 4, 2023, the Thai Food and Drug Administration (Thai FDA) initiated a 24-day public consultation, unveiling four draft notifications aiming at revising the cosmetic ingredient lists—a crucial set of technical standards for cosmetics in the country. The objective is to align these regulations with the updated ASEAN Cosmetic Directive (ACD). 1
The proposed changes encompass the addition and revision of permitted preservatives, as well as restricted and prohibited ingredients in cosmetics. Specifics are as follows (the text in red indicates changes to the previous lists):
1. List of Prohibited Ingredients: one added and one revised 2
Status | Ref. No. | Substances | CAS No. |
Revision | 343 | Dioxane, except for contamination in the finished products that does not exceed 10 parts per million by weight (10 ppm or 10 mg/kg). | 123-91-1 |
Addition | 1611 | 4-[(tetrahydro-2H-pyran-2-yl)oxy]phenol (Deoxyarbutin, Tetrahydropyranyloxy Phenol) | 53936-56-4 |
If cosmetic stakeholders will have already manufactured, imported, or sold products containing the newly prohibited ingredient Deoxyarbutin (No. 1611) before the effective date of the official announcement, they will be granted a 180-day grace period.
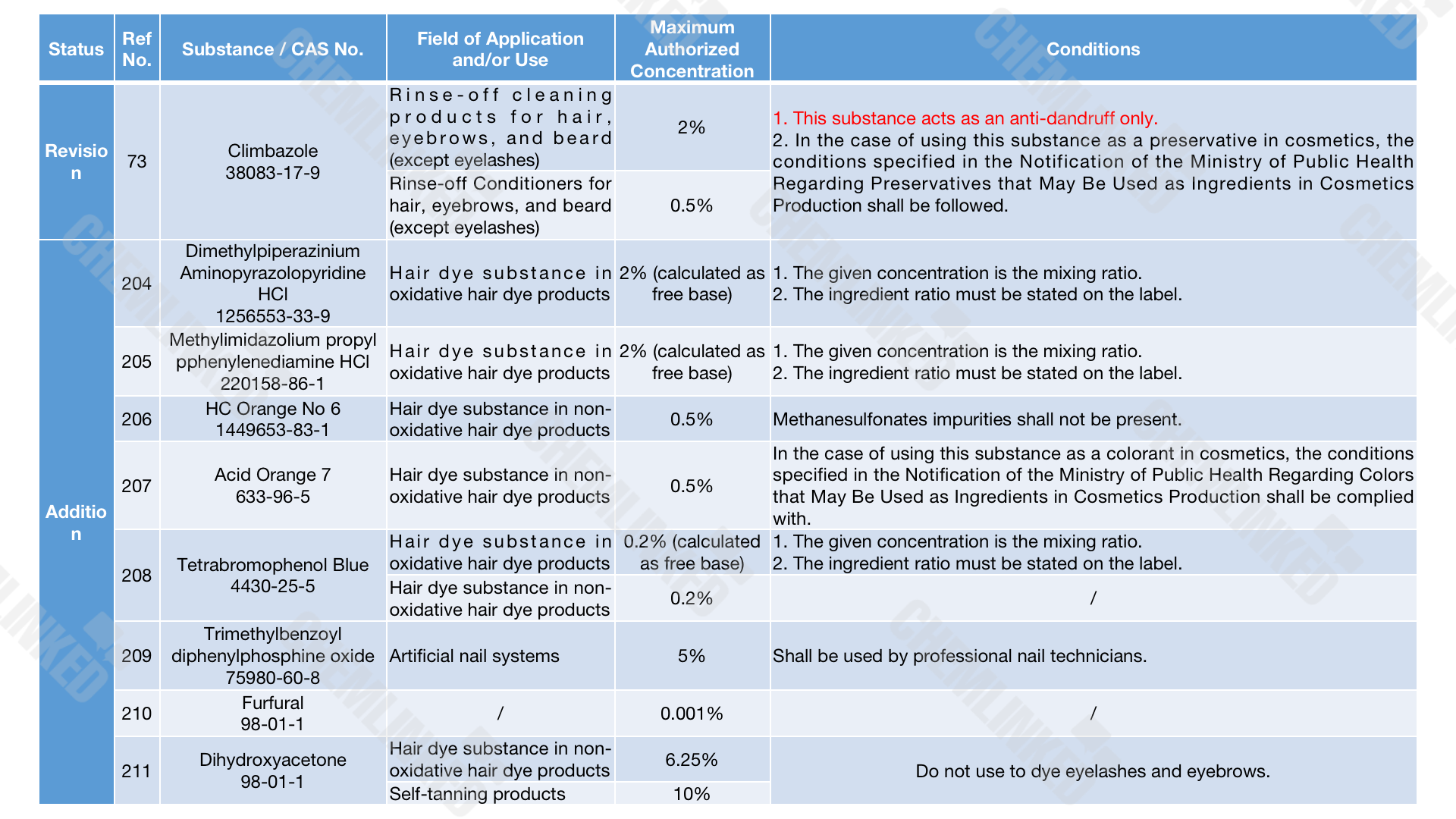
2. List of Restricted Ingredients: eight added and one revised 3
With reference to updates in the ACD, the Thai FDA intends to amend the use requirements for Climbazole and introduce eight new restricted ingredients. Similar to the grace period for prohibited ingredients, cosmetic stakeholders will have 180 days after the effective date if they have already manufactured, imported, or sold items containing the listed ingredients.
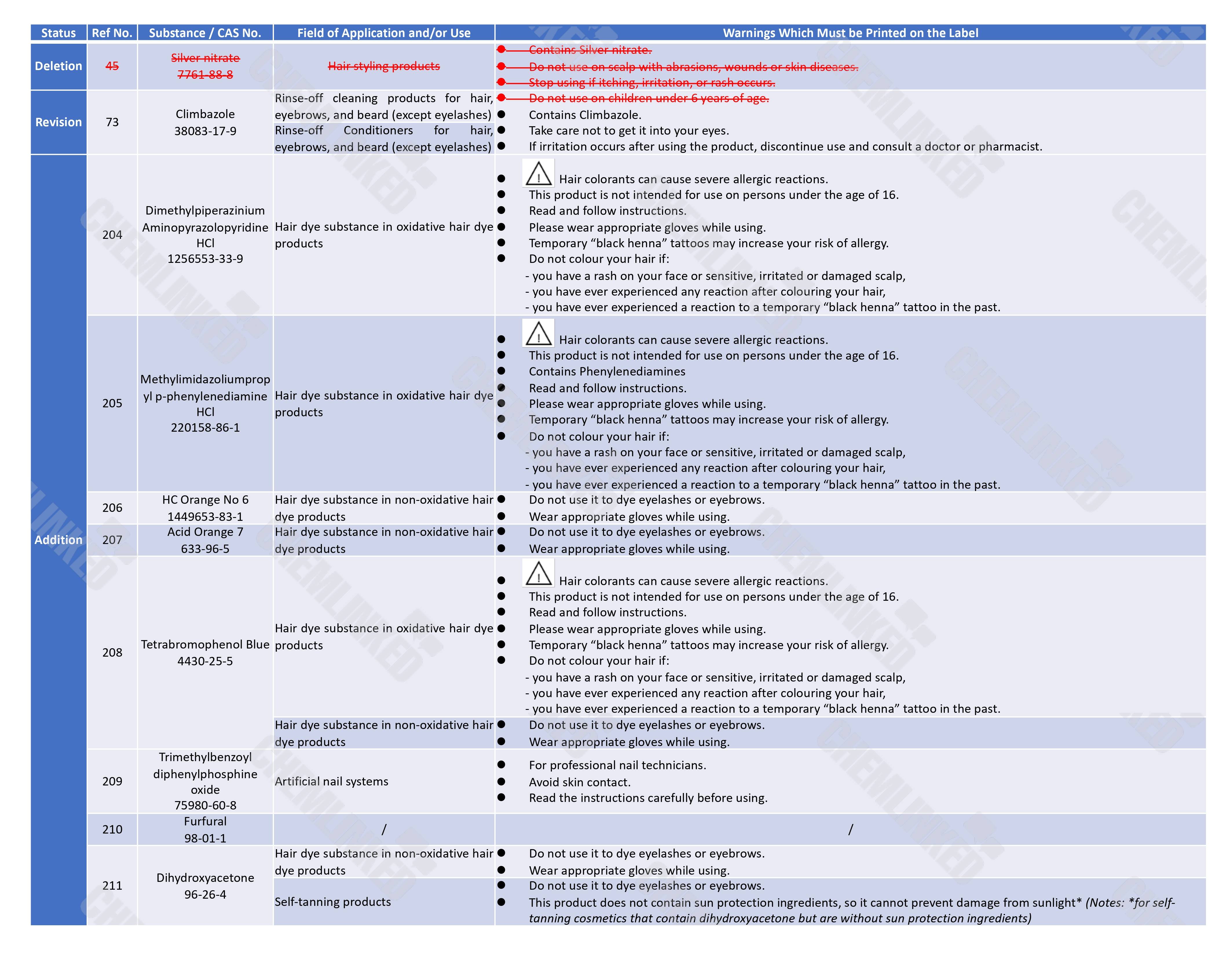
Labeling Updates 4
Moreover, to maintain consistency with the revised restricted ingredient list, the Thai FDA also plans to modify the labeling requirements for the affected ingredients in the List of Warnings on Labels for Cosmetic Containing Restricted Ingredient. The labeling requirements for silver nitrate (original entry 45), currently listed as a prohibited ingredient, will be removed. Additionally, the labeling requirements for Climbazole (No. 73) will be modified, and the labeling requirements for the eight newly listed ingredients will be added.
Products with old labels can be sold in the transitional period but must with updated labels that comply with the new requirements within 180 days after the official effective date.
3. List of Permitted Preservatives: one revised 5
Status | Ref No. | Substance / CAS No. | Maximum Authorized Concentration | Conditions |
Revision | 30 | Climbazole 38083-17-9 | 0.5% |
|
0.2% |
|
It is important to note that if cosmetic stakeholders will have already manufactured, imported, or sold products containing Climbazole before the effective date of the official announcement, they will also enjoy a 180-day grace period.




 Request a Demo
Request a Demo
 We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










