On Sept. 10, 2019, China National Medical Products Administration promulgated a new testing regulation - Working Rules for Cosmetic Registration and Filing Testing (hereinafter referred to as the "the Working Rules") to optimize the previous testing system and improve the testing efficiency of cosmetic registration and filing.
The new rules have triggered hot discussion among the industry of how to better adapt to the new testing system. This article will focus on analyzing regulatory changes and their impact on testing institutions and cosmetic manufacturers.
Background: Administrative Reform
In China, one of the most crucial market entry prerequisites facing imported cosmetics is filing or registration and involves entrusting a testing agency to conduct technical tests and generate a qualified report with sufficient data to substantiate the safety and efficacy of products. As per the Measures for Cosmetic Administrative Licensing Testing and Measures for Qualification of Cosmetic Administrative Licensing Testing Institutions, the cosmetic testing institutions for administrative license shall be accredited by the NMPA and the testing institutions for filing shall be designated by the provincial MPAs.
Up to now, China has appointed over 200 cosmetic filing testing institution and has accredited 35 testing agencies for cosmetic registration, 28 of which are hygiene safety testing institutions and the rest 7 are 3A hospitals responsible for human clinical and safety testing. In addition, the distribution of testing resources is unbalanced across the country. In the past the technical and staffing capacities of these institutions have been overburdened by the extensive testing workload which has translated to major bottlenecks in cosmetic registration and filing work.
The Working Rules were developed to alleviate these issues and address the contradiction between an increasing demand for registration and filing testing and the limited testing resources under the current regime.
Significant Changes to the Working Rules
Consisting of 24 articles and two annexes, the Working Rules specify the testing procedures for cosmetics registration and filing, management requirements of testing institutions, testing item requirements for different types of products as well as the templates and styles of testing reports. Key amendments to the working rules are summarized below:
1. Terminate the original designated testing institution system
The authorization system of testing institutions has been officially canceled. Relevant qualifications of cosmetic testing institutions previously sanctioned by the former CFDA will be automatically terminated starting from November 1, 2019.
As per the Working Rules, testing institutions can undertake testing work if the following qualification criteria are satisfied from September 10, 2019.
Have an independent legal entity;
Have obtained China Metrology Accreditation (CMA) and the business scope of its CMA can meet the needs of cosmetics registration and filing testing work (except for testing items that have not been included in the scope of CMA certification);

Testing institutions engaged in human safety and efficacy evaluation testing of cosmetics should also have two or more full-time staffs with the practitioner qualification certificate of dermatologically related major and more than 5 (including 5) years of working experience in cosmetic safety and efficacy assessment. The trial subjects’ informed consent system, volunteer management system and adverse reaction management system shall also be established by these institutions.
Have submitted all required information through the online testing information system.
2. Simplify the workflow of cosmetic testing
Another major change is the implementation of digital information management. China NMPA has newly established online testing information system to replace the previous offline application of cosmetic testing that was handled by the receiving window of testing institutions.
(Online testing information system)
This action likely foreshadows the inevitable integration of the testing information system, the cosmetics registration management system and the filing management system, which will allow whole process management of cosmetics registration and filing and ensure the quality and safety of cosmetics.
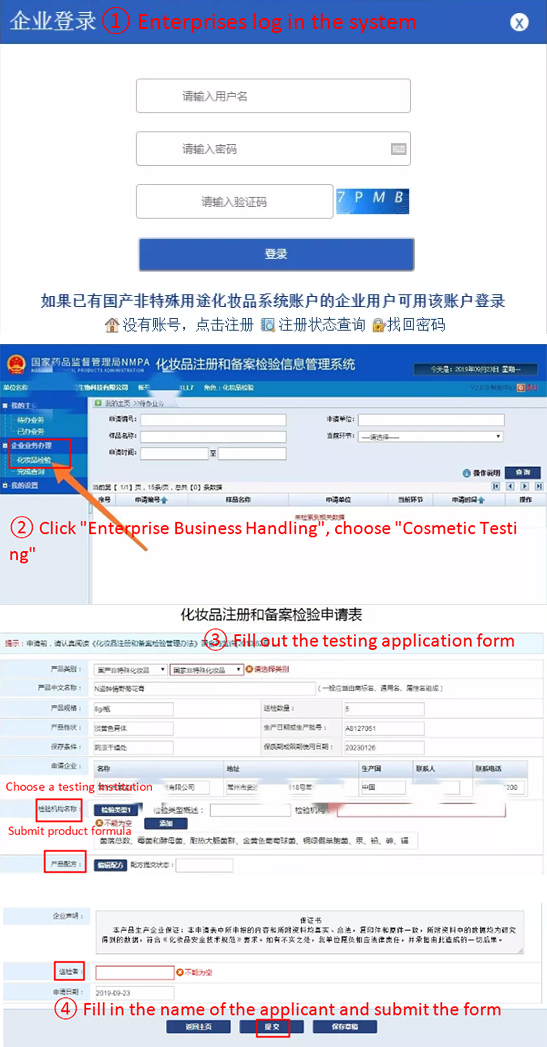
The general procedures of online testing application are shown below (take the test application of domestic non-special use cosmetics as an example [4]):
(Source: Shiputest)
During the current transitional period running from September 10, 2019 to October 31, 2019, cosmetics enterprises can either select testing institutions that have published relevant information on the testing information system to apply for cosmetic testing or follow the original procedures. However, starting from November 1st, 2019, cosmetics stakeholders shall only carry out cosmetic registration and filing testing via the online testing information system.
3. Standardize the requirements of testing items
In addition, the new rules have changed the original test items for cosmetics registration and filing. Specific changes are as follows:
(1) The microbiological test item remains unchanged.
(2) Updates to the physicochemical test items
For products that claim to contain alpha-hydroxy acid or that does not claim to contain alpha-hydroxy acid but its total amount is higher than or equal to 3% (w/w), both the alpha-hydroxy acid item and the pH value shall be tested.
The Working Rules newly increase the dioxane (containing ethoxy structural ingredients) and free formaldehyde test items. The two items shall be tested in the same laboratory.
|
Pre-market testing of anti-acne products will no longer include tests for antibiotics such as metronidazole. This will be tested during post-market sampling and testing.
For finished products that cannot be sampled or may affect the testing results due to packaging reasons (such as spray products, air cushion products and etc.), the enterprises shall also provide semi-finished products (finished to state just before the final packaging process) when submitting the test samples, which shall be clearly stated by the testing institution in the report.
(3) Updates to the toxicological test items
For products with a chemical sunscreen content higher than or equal to 0.5% (w/w) (except for rinse-off, perfume, and nail polish products), skin phototoxicity test and skin sensitization test should also be performed.
Bath products should be tested for acute eye irritation.
Non-oxidative hair dye products are not required to conduct the bacterial reverse mutation assay and in-vitro mammalian cell chromosomal aberration test.
(4) Updates to the human safety test items
When conducting the skin patch test for freckle-removing and sunscreen cosmetics, if the result shows irritation or the result is difficult to judge, then arepeated skin open application test should be added.
Rinse-off products that claim anti-acne, anti-wrinkle, and freckle-removing, etc. should have their clinical efficacy and safety substantiated through human trials.
(5) Updates to the efficacy evaluation test items
Products which claim freckle-removing/whitening, anti-hair loss, and claim new efficacy should be tested according to the test items requirements determined by the Cosmetic Efficacy Claims Evaluation Guidance. However since the Guidance has not been implemented yet, efficacy evaluation testing is currently not possible.
4. Test sample requirements
According to the Working Rules, all test samples should be provided to the first testing institution that accepts the application. The number of required samples is determined by the testing agency according to individual inspection and sample retention requirements. Submitted samples shall have a complete package and Chinese label marked with information pertaining to registration and filing testing such as the product name, the manufacturer’s name, the production date and shelf life or the batch number and expiration date.
In cases where imported special-use cosmetics have submitted test samples forregistration testing, one sample sealed by the testing agency and one unopened market product shall be submitted.
In cases where the test samples are inconsistent with the marketed products, the registration or filing test report of the product shall be invalidated and stakeholders shall be punished for submission of false registration materials or false filing.
Impact for Cosmetic Enterprises
Given the Working Rules put forward a lot of new requirements for cosmetics testing institutions and enterprises, a grace period was granted to ensure a smooth transition. Transitional policies include:
For tested cosmetics or cosmetics which have completed sunscreen testing items and obtained the testing report from overseas laboratories, these test reports can continue to be used in the registration or filing of the products.
For products that have been registered or filed and are in a situation where the original test items are inconsistent with the “Working Rules”, they shall complete the additional test items within one year after the issuance of this notice, in line with the test items requirements (excluding human safety test items) specified in the “Working Rules”. The supplementary report shall be submitted when applying for extending the validity period of product administrative license or provided for future reference when confirming through the online filing system that the filed products will continue to be produced.
For cosmetics which have already applied for registration or filing but have not yet been tested, the testing shall be carried out and the testing reports shall be issued in accordance with the requirements of the “Working Rules” from September 10, 2019.
In all, the implementation of the Working Rules allows more third-party testing agencies to participate in the test work for cosmetics registration and filing, helping streamline the administration, and hopefully alleviate the backlog of enterprises awaiting testing services under the old system.
Additionally, two of the most significant changes bear repeating. First, the marketed products must be consistent with the samples submitted for testing otherwise enterprises will be subject to punishment. Second, given the test items for certain cosmetic categories such as sunscreens, anti-wrinkle, anti-freckle, anti-acne, spray, air cushion product has increased significantly it follows that testing costs and time required to complete testing will also increase accordingly.
For further cosmetic testing compliance consultancy, please contact [email protected].




 Request a Demo
Request a Demo
 We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 









