From 2008 to 2012, 4 years has witnessed merely 3 new cosmetic ingredients (NCIs) approved by SFDA (now CFDA) in spite of the continued submission of application dossiers. According to statistics from CFDA, more than 130 dossiers have been submitted for the administrative licensing of NCIs, while most of which are never heard back since.
What are major stumbling blocks that make the registration process extremely challenging and drawn-out? The cosmetics registration expert, Tommy Kong, who is also proficient in new chemical notification in China, has compiled the "Guidance on Pre-market Approval of New Cosmetic Ingredients in China", which is intended to throw light upon key areas that cosmetic manufacturers need special attention, including determination of a NCI, dossiers of vital importance, safety criteria and evaluation, exemption cases of toxicological data as well as other highlights and advices.
The general procedures would be quite clear and simple as shown below:
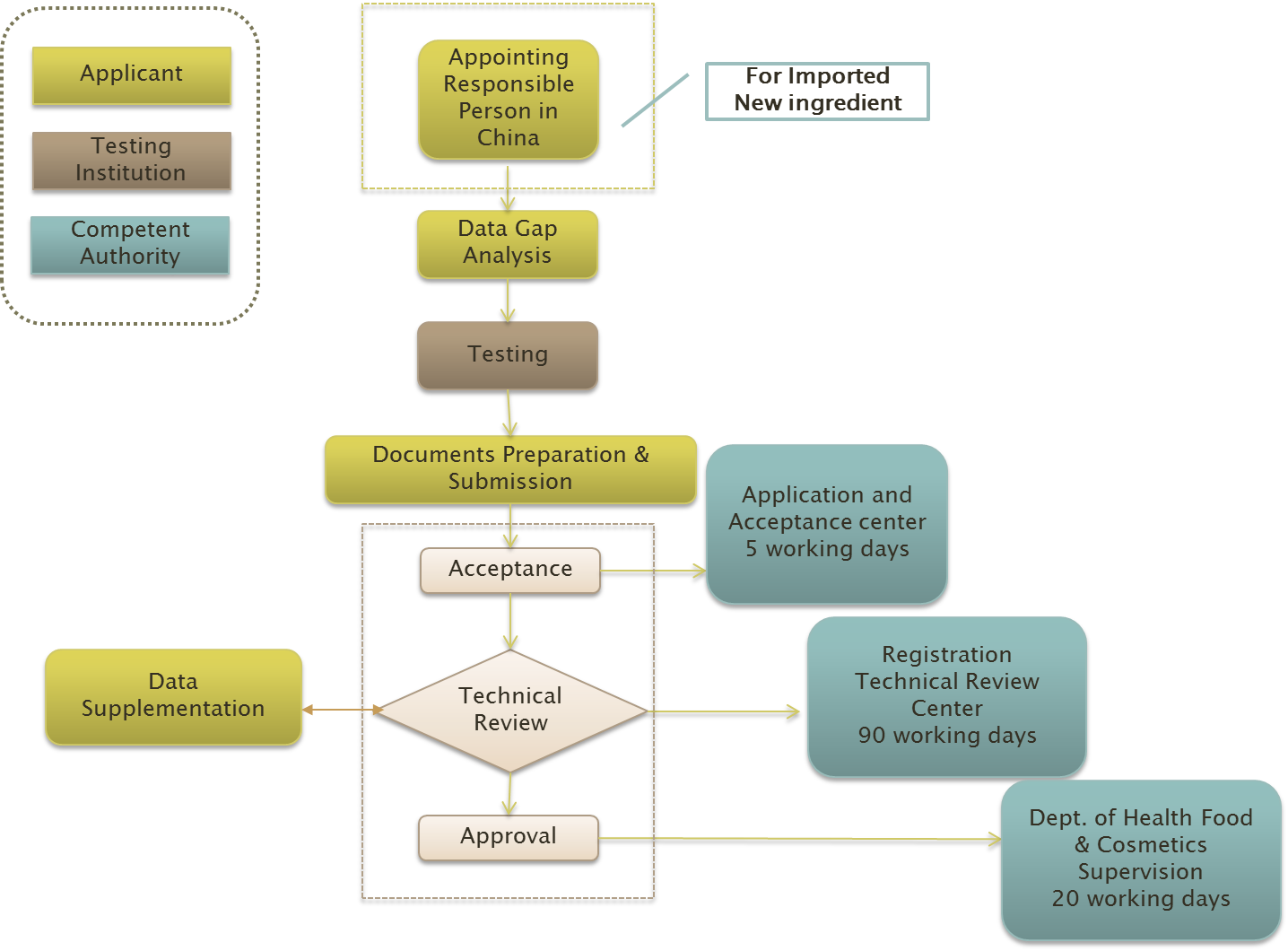
However, to determine whether an ingredient is new or not could be obscure. In principle, there are three golden criteria according to Tommy's Guidance:

Even with these criteria, companies may not succeed in figuring out the status of the ingredient. Firstly, IECIC 2003 is an internally circulated list and IECIC 2013 is still on its way to be finalized, just two batches of which have been released by CFDA (see Chemlinked news on 14 May).
Assuming that an ingredient is not included in the IECIC, the next step is to check whether the ingredient has been used in licensed special use cosmetics, but the lack of a full list of those products has made this task tough. In case that the ingredient is plant extract, problems still occur to make the identification confusing. For example, the nelumbo nucifera gaertn is an approved plant, therefore, its extracts are also deemed as existing ingredients which is also true for its fruit extract, nelumbo nucifera seed powder.
Application dossiers must be prepared as complete as possible, especially the R&D report, specification and quality control data, toxicological safety evaluation data, etc. It is mandatory to provide the quantitative/ quantitative methods and other analytical methods for quality control or impurities identification. Once the testing methods are self-developed and not officially recognized, verification test would have to be performed in CFDA-accredited testing institutions to prove their scientific basis, emphasized by Tommy in his Guidance .
The toxicological safety evaluation data should be given the priority concern, including a summary of the evaluation, required toxicological data and safe data on potential safety risk substances. Nevertheless, practical preparation could still be demanding due to the absence of safety evaluation methodology for ingredients. In such circumstances, the potential applicants can resort to relevant guidelines from Scientific Committee on Consumer Safety and other authorities.
There is a list of tests that are required for the registration of a new cosmetic ingredient, but in certain cases, some of which can be exempt, according to the Guidance on Application and Review of New Cosmetic Raw Materials issued by CFDA in 2011:

Tommy also highlighted that the CFDA expert panel is very concerned with the potential presence of impurities so the applicant must be capable of fully describing impurity identities, contents, physical and chemical parameters, shelf life, storage conditions and so on.
To deal with such a technically demanding registration, companies are strongly recommended to assign highly qualified regulatory affairs expert or an agency to assist you to achieve the mission that seems to be impossible.
Reference Links
-
[China Cosmetics Guidance in a Nutshell] Volume 2: Pre-market Approval of New Cosmetic Ingredients in China (For Free Download)
Guidance on Application and Review of New Cosmetic Raw Materials




 Request a Demo
Request a Demo We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










