In the past few years, rumors have that China is intended to lower the regulatory barrier for cosmetic registration and alleviate the heavy pre-market application burden of cosmetic companies but to intensify the post-market supervision and increase enterprises’ sense of responsibility for safety of commercially-available cosmetic products.
Recently, in the main duties and staffing of CFDA (see Chemlinked news on 22 May), which was announced on 15 May 2013, two great changes that might affect cosmetics industry were observed:
Administrative licensing
The manufacturers of imported non-special use cosmetics (Non-SUC) are no longer required to register their products with CFDA but only with the food and drug administrations at the provincial level. The procedure will be similar to that of domestic non-special use cosmetics.
The modification for cosmetic licensing is presented in the table below:
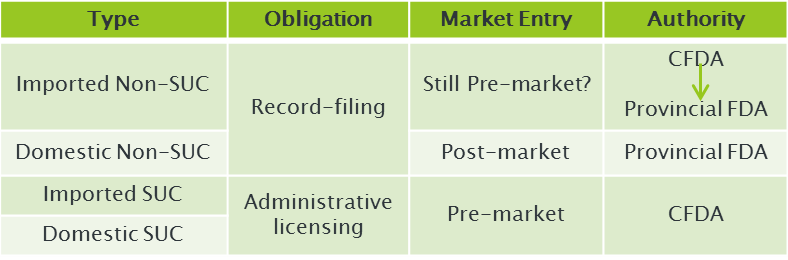
Previously, quite a few complaints have been received from many foreign cosmetic companies and industrial associations about Chinese government’s differential treatment towards imported and domestically-made non-SUCs.
Even for the same kind of products, imported ones are subject to pre-market registration with CFDA while domestic ones only need to obtain a record-filing certificate from the provincial FDA within two months after cosmetic products are placed on the market. It is expected the devolution of power this time will to a certain extent ease the dissatisfaction and will definitely shorten the registration duration, said by Martin Hu, a cosmetics regulatory expert from REACH24H Consulting Group.
Mr Hu also pointed out that the detailed guidance on the registration of imported ordinary cosmetic products is pending so it remains unknown whether the pre-market application will still be required. Meanwhile, the overseas manufacturers may be faced with inconsistent approval and supervision requirements carried out by different food and drug administrations at provincial level.
Mr. Tommy Kong from REACH24H who attended the Academic Meeting organized by the Chinese Society of Toxicology recently told Chemlinked reporter that the specific implementation plan is under discussion. The government has to take into account of discrepant resources or abilities that provincial FDAs possess. Obviously, FDAs in coastal areas will receive more application cases and encounter more problems than those in less developed regions. Therefore, several competent centers are considered to be established to allocate and coordinate resources, to jointly deal with challenges and to enhance the supervision.
While the good news is that CFDA experts can be more concentrated on the review of special use cosmetics and new cosmetic ingredients for more efficient and transparent registration procedures.
Production Licensing
Another big change is the CFDA takes over the responsibility of manufacturing licensing of cosmetic products from AQSIQ. Previously, cosmetics production was under dual regulation of CFDA and AQSIQ. Cosmetics manufacturers in China needed to obtain the Hygiene License for Production Enterprise of Cosmetics from Provincial FDAs and the Cosmetics Production License from provincial branches of AQSIQ. The overlap between the two similar licenses was an extra burden for cosmetic producers. According to Ms Feng from China Association of Fragrance Flavor and Cosmetic Industries (caffci), the two licenses are to be integrated into one license and overlapping requirements will be cancelled.
Under the new cosmetic roduction licensing system, manufacturers who produce cosmetics in China now only need to apply for the Hygiene License (or any modifications to this license) from provincial FDAs. But it is still not sure when and how provincial FDAs will implememt the policy change now. Insiders predict major economic hubs, such as Shanghai, Guangzhou, Zhengjiang and Jiangsu, where most of cosmetics producers are located will take the lead.
Reference Link
- CFDA news release: CFDA Main duties and staffing
- [China Cosmetics Guidance in a Nutshell] Volume 1: Steps to Exporting Cosmetic Products in China (free download)




 Request a Demo
Request a Demo We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 








