On May 22, 2022, the Saudi Food and Drug Authority (SFDA) issued the Guide for Cosmetics Containing Musk and Ambergris (the Guide) 1 to instruct international stakeholders on the requirements and conditions for manufacturing and trading cosmetics with musk and ambergris in the Kingdom of Saudi Arabia.
The Guide, which will come into force on November 2, 2022, specifies the definitions and types of natural musk and natural ambergris, excluding active substances that are chemically prepared to imitate the natural odor of musk and ambergris. The Guide applies to perfumes and cosmetics containing musk or ambergris fragrance extracts.
These products shall be prepared and manufactured in accordance with Cosmetics Good Manufacturing Practice to ensure product safety. And these products shall be subject to laboratory tests, including microorganisms, heavy metals, impurities, phthalates, nitro musks, allergens, and other items. 2
Among them, the microbiological testing items include the total count of aerobic mesophilic bacteria, pseudomonas aeruginosa, Escherichia coli, staphylococcus aureus, yeast and mould, candida albicans, etc.
 The microbiological testing items
The microbiological testing itemsIn the phthalates testing items, the following 11 prohibited phthalates shall not be detected.
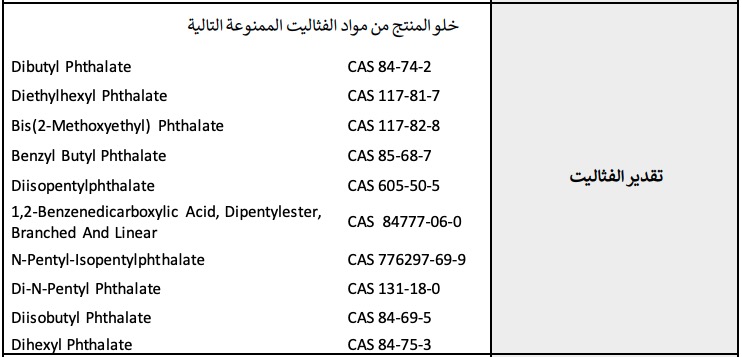 The phthalates testing items
The phthalates testing itemsIn the nitro musks testing items, Musk Ambrette (CAS 83-66-9) and Musk Tibetene (CAS 145-39-1) are banned substances. 2




 Request a Demo
Request a Demo
 We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










