On June 9, the Hebei Medical Products Administration (Hebei MPA) issued an internal notice clarifying the filing requirements for non-special use cosmetics whose product name is “original liquid” (“原液” in mandarin Chinese)[1]. Cosmetic filing and regulatory departments at all levels within the province are required to carry out the filing and supervision work of related products in line with the requirements.
Specifics are as follows:
Liquid personal care cosmetics that are composed of a single component or a single category of components as a functional ingredient, mixed with necessary solvents, antioxidants, preservatives, and used directly or diluted according to a prescribed ratio, can use the "original liquid (原液)" as the product attribute name.
Products using “original liquid” as the attribute name should clearly identify the target body parts (skin or hair use) in the instructions for use or be used together with the common name of "skincare" or “hair care."
Relevant industry insiders revealed that the Wuxi (the prefecture-level city in Jiangsu Province) Medical Products Administration also circulated this document and requested enterprises’ compliance with the specified two requirements.
The Status Quo of Original Liquid Cosmetics
Booming Market
In recent years, spurred by consumers’ continuing quest for natural, safe, and effective cosmetic ingredients, the “original liquid” cosmetics featured single-component and precise positioning as a remedy to specific skin problems burst into the public consciousness. According to the 2018 Hot Trends Report on China's Fashion Beauty, the original liquid products containing a single chemical formula are welcomed by consumers. The annual growth rate of online consumption exceeds 50% [2].
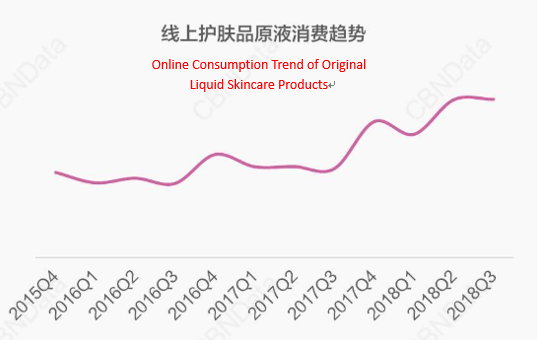 (CBNData)
(CBNData)
No Oversight
The growing market has spawned a lot of chaos as there has been no official conclusion on the definition of “original liquid” cosmetics as well as no regulatory standards for a long time. In fact, the industry's common understanding is essentially the same as what is stated in the above notice—the product refers to a diluent containing only a single component. But in the eyes of consumers, the “original liquid” product is often compared with an essence. It is considered a product with a single active ingredient and a higher concentration, that is, a product with high potency.
Some enterprises exploited regulatory loopholes and consumers’ strong urge to purchase high efficacy products and produced plenty of substandard products. Those so-called "original liquids" are not cosmetic products with simple ingredients as expected by consumers. In addition to the necessary solvents, antioxidants, preservatives, moisturizers, emollients, film-forming agents, etc. are also added. According to one cosmetic R&D specialist, for these type of products "There is no obvious difference between these “original liquid” products and essence. It is completely a “marketing idea” used by some cosmetic manufacturers."
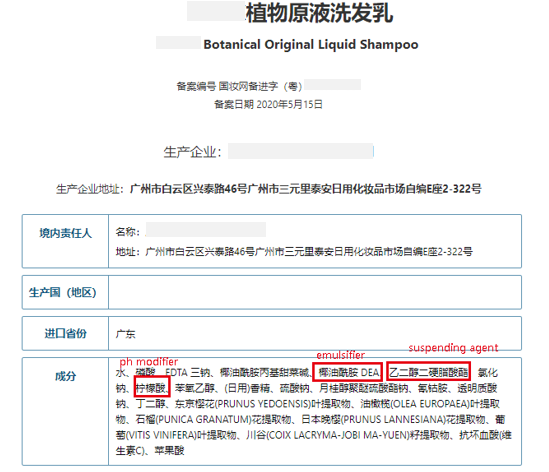 (NMPA)
(NMPA)
Implication for Stakeholders
The Hebei Province notice is of considerable significance as it puts forward the precise definition and concentration requirements for the first time. However, it shall be noted that the document does not highlight "high concentration"; it only indicates "used directly or diluted according to a prescribed ratio". Its implementation will overhaul and standardize the "original liquid" market in Hebei and provides a reference for the filing of “original liquid” products in other areas.




 Request a Demo
Request a Demo
 We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 








