1. Definition & Classification of Quasi-drugs
Under the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices, Japan legally classifies cosmetics into two categories: cosmetics and quasi-drugs. The most significant difference between quasi-drugs and cosmetics is whether they contain “active ingredients” that claim specific effects.
The quasi-drugs are defined as "substances which have mild effects on the human body, and have a specific function other than drugs or medical devices." There are mainly the following three categories:
No | Scope | Products Example |
Ⅰ | (a) Products for preventing nausea and other discomforts, or those for preventing bad breath or deodorizing the body; (b) Products for preventing heat rash, sores, etc.; (c) Products for preventing hair loss, or those to promote hair growth or remove hair; | Oral freshener, Deodorant, Antiperspirant, Baby powder, Depilatory, Hair growth products, etc. |
Ⅱ | (d) Products which used to exterminate mice, flies, mosquitoes, fleas, or other animals or insects similar to these for the benefit of the health of humans and animals; | Helminthic |
Ⅲ | (e) Products designated by MHLW containing approved active ingredients that have pharmacological action | Medicated cosmetics (including anti-dandruff products, whitening products, and anti-acne products), hair dyes, hair perms, medicated oral-care products, etc. |
2. Management Requirements for Quasi-drug Ingredients
Japan implements an approval system for quasi-drugs. The focus of the approval lies on the efficacy, safety and quality specifications of ingredients. At present, quasi-drugs’ ingredients are subject to the management of two kinds of lists:
1)Lists published by the MHLW:
Enterprises can only use the ingredients in the following lists to produce quasi-drugs, and the concentration and specifications of the ingredients used must be consistent with those specified in the lists.
Japanese Standards of Quasi-drug Ingredients 2006 (JSQI 2006)
Since 2006, the Japanese Standards of Quasi-drug Ingredients 2006 has undergone nearly 16 revisions with the latest amendments to JSQI 2006 made on Sept. 11, 2020 [1].
The current JSQI 2006 lists all the active ingredients that are permitted to be used in quasi-drugs. Enterprises could use the JSQI 2006 to find the information about the ingredients, which included information such as the properties, confirmation tests, purity tests, quantitative methods etc.,
For example, the pyrogallol that can be used for hair dyeing. The JSQI 2006 stipulated the property, confirmation test, melting point, purity test, loss on drying and ignition residue.

Notably, under certain conditions, enterprises can use alternative methods other than those specified in JSQI. According to FAQs about Standards for Cosmetics and the Manufacturing and Marketing Approval of Quasi-drugs released by MHLW, if there are multiple test methods for the same test purpose, priority should be given to the test method specified in JSQI. But, if there is an alternative method that the reproducibility and accuracy are not inferior to the prescribed test method, the alternative method can also be used.
However, the JSQI 2006 only specified the basic information for the quasi-drug’s ingredient. Enterprise who wants to know the specific use limits of the permitted active ingredient in different categories of quasi-drug products should refer to the following four lists:
1. Permitted Active Ingredients in Hair Dyes[2]
This list displays the active ingredients which are permitted to be used in hair dyes products, and their corresponding usage.
2. Permitted Active Ingredients of Permanent Wave Products[3]
This list includes two tables. Table 1 shows the classification of active ingredients of Permanent Wave Agents. Table 2 displays the active ingredients with their permitted range as well as the upper limit for the content of active ingredients and the oxidizing activity per dose per person.
3. Permitted Active Ingredients in Bath Additives[4]
Column | Ingredient Standard | Ingredient Name | Combination Concentration (%) |
I | P, Q,F | Potassium Chloride | 1.0-99.0 |
I | P, Q | Sodium Chloride | 1.0-99.0 |
I | F | Magnesium Chloride | 1.0-99.0 |
I | Q | Sodium Sesquicarbonate | 1.0-99.0 |
I | P, Q, F | Sodium Bicarbonate | 1.0-99.0 |
I | F | Sodium Carbonate (anhydrous) | 1.0-99.0 |
I | F | Sodium Carbonate (crystal) | 1.0-99.0 |
I | P | Sodium Carbonate Hydrate | 1.0-99.0 |
I | P | Dried Sodium Carbonate | 1.0-99.0 |
I | P | Sodium Thiosulfate Hydrate | 1.0-99.0 |
I | Q | Anhydrous Sodium Thiosulfate | 1.0-99.0 |
I | Q | Sodium Sulfate | 1.0-99.0 |
I | Q | Exsiccated Sodium Sulfate | 1.0-99.0 |
I | F | Sodium Sulfate (anhydrous) | 1.0-99.0 |
I | F | Magnesium Sulfate (crystal) | 1.0-99.0 |
I | F | Magnesium Sulfate (dried) | 1.0-99.0 |
II | P | Potassium Bromide | 2.0-4.0 |
II | F | Calcium Carbonate | 1.5-10.0 |
II | Q | Precipitated Calcium Carbonate | 1.5-10.0 |
II | P | Precipitated Calcium Carbonate | 1.5-10.0 |
II | Q | Alum | 1.0-20.0 |
II | P | Aluminum Potassium Sulfate Hydrate | 1.0-20.0 |
II | Q | Exsiccated Alum | 1.0-20.0 |
II | F | Aluminum Potassium Sulfate(dried) | 1.0-20.0 |
II | P | Ferrous Sulfate Hydrate | 0.05-20.0 |
Active ingredients that must be used are listed in column I of the table and those listed in the column I of the table must constitute 70% or more of the bath additive. Any single active ingredient listed in column I of the table must constitute 25% or more of the bath additive. Each active ingredient must meet the standards in accordance with the following Japanese standards abbreviated as: · “P”: Pharmacopoeia; · “Q”: Japanese Standards of Quasi-Drug; · “F”: Japan’s Specifications and Standards for Food Additives. | |||
4. Permitted Active Ingredients in Medicated Cosmetics[5]
This list specifies the names and specifications of active ingredients contained in the following 8 categories of medicated cosmetics.
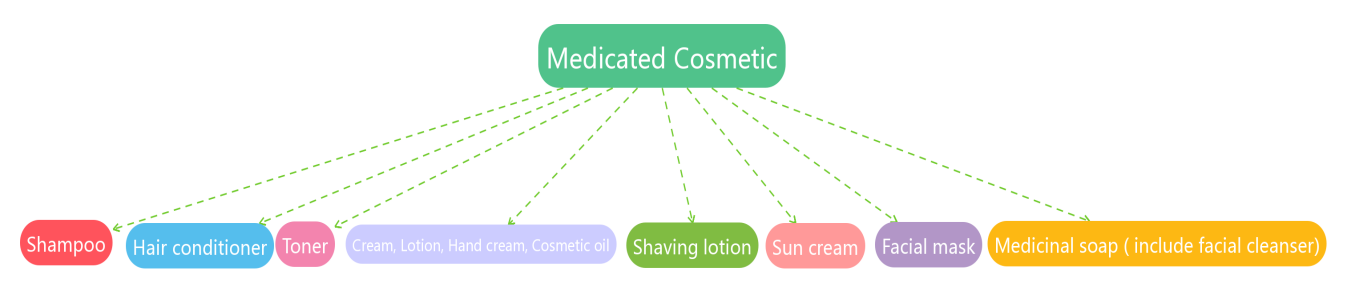
The specification requirements are mainly determined by The Japanese Pharmacopoeia, The Japanese Pharmaceutical Codex 2002 and JSQI 2006. Enterprises need to pay attention to the fact that the specifications of different active ingredients in different kinds of quasi-drug products may be different.
For example, the following table is a part of the Permitted Active Ingredients in Medicated Cosmetics. The Sulfur content of hair conditioner is only 10% of that in shampoo; the proportion of Salicyclic Acid in shampoo is 0.1-2, but for hair conditioner it’s 0.3; the proportion of Pyrithione Zinc in shampoo is 0.3-1, while that in hair conditioner is 0.3-0.75;
Categories of Medicated Cosmetics | Ingredient Name | Specification and addition | ||
Japanese Pharmacopoeia | Japanese Pharmaceutical Codex 2002 | JSQI 2006 | ||
Shampoo | Sulfur | 5 | ||
Dipotassium Glycyrrhizinate | 0.1 | 0.1 | ||
Salicyclic Acid | 0.1~2 | 0.1~2 | ||
Pyrithione Zinc | 0.3~1 | 0.3~1 | ||
Hair Conditioner | Sulfur | 0.5 | ||
Isopropyl- para-cresol | 0.1 | |||
Salicyclic Acid | 0.3 | 0.3 | ||
Pyrithione Zinc | 0.3~0.75 | 0.3~0.75 | ||
List of permitted additives in quasi-drugs[6]
In Japan, ingredients other than active ingredient that are allowed to be used in quasi-drugproducts are regarded as additives, and the MHLW manages these ingredients by formulating the “List of permitted additives in quasi-drugs”.
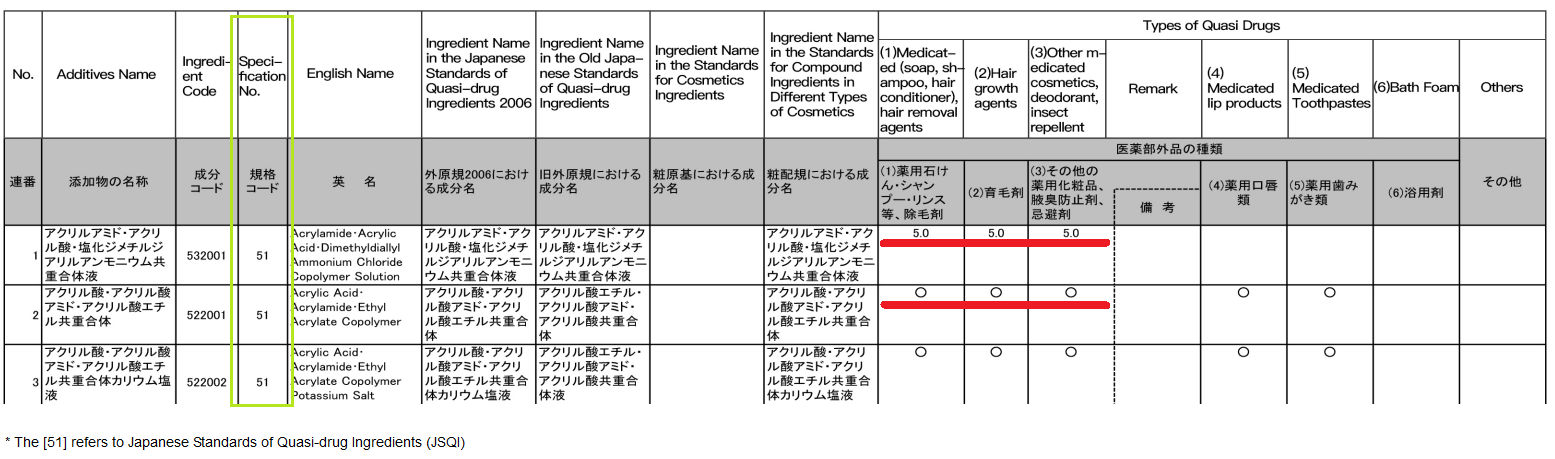 This additive list clearly specifies the specification and application scope of additives. The specification indicated in the list generally has the following two forms.
This additive list clearly specifies the specification and application scope of additives. The specification indicated in the list generally has the following two forms.
(1) Specifications Marked with Specific Value:
For quasi-drug additives marked with specific specifications, the content of the additive in quasi-drugs must be within the value specified in this list.
(2) Specifications Marked with a Circle (○):
The circle indicates that the specification of the quasi-drug additive is provided. Enterprises shall refer to the “Specification No.” Column of the List for more details. For example, the 51 in the list refers to the Japanese Standards of Quasi-drug Ingredients 2006.
In addition, if the ingredient marked with a circle (○) in the "Types of Quasi Drugs" column is blended as an additive for the corresponding quasi-drug, there is no need to submit safety certification materials while applying for approval in principle.
However, if the additive has an effect like an active ingredient based on the actual dosage, the applicant may be requested to explain.
2)Lists of patented quasi-drug ingredients of enterprises:
This kind of permitted ingredients lists for quasi-drugs are separately applied by enterprises and approved by MHLW. Besides, due to the protection of property rights, this list will not open to the public and is controlled by MHLW. It is worth mentioning that the ingredients on the list are new ingredients for other enterprises.
3. Management Requirements for New Quasi-drug Ingredients
Generally, ingredients outside the permitted list are regarded as new ingredients. However, there is a case that although the ingredients are listed as permitted ingredients, they will be considered as new ingredients if they use concentration or specifications exceeding the limits specified in the list.
The MHLW implements a strict approval system for new ingredients. To use new ingredients or expand the scope of use (the use concentration or specifications exceed the regulations), the enterprises must first declare the new ingredient.
When an enterprise declares a new ingredient, it shall provide information on the use background, physical and chemical properties, safety and stability. It should be pointed out that the approval is not only for ingredients, but also involve ingredients’ scope of use, dosage and specifications.
4. Suggestions for Stakeholders:
If the additive in quasi-drug acts as an active ingredient based on the actual dosage, applicants need to prepare explanations for it. Therefore, enterprises need to strictly obey the provisions of the list of ingredients for quasi-drug products, reasonably and effectively use the permitted active ingredients and additives to produce quasi-drug products.
The approval of the new ingredients is not only depended on the ingredients itself but also the scope of use, dosage and specifications. Once the enterprise finds that the use of ingredients has changed during manufacturing, it should re-declare it immediately.




 Request a Demo
Request a Demo
 We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 










