On February 1, 2023, EU Scientific Committee on Consumer Safety (SCCS) released the final opinion on alpha-arbutin (CAS No. 84380-01-8) and beta-arbutin (CAS No. 497-76-7).1 Compared to the previous respective opinions on these two ingredients, the potential combined use of the two ingredients in cosmetics are taken into consideration in the latest opinion. Besides, the hydroquinone contamination requirements for their applications are further clarified.
Commonly serving as antioxidant, skin bleaching agents and skin conditioning agents, alpha-arbutin and beta-arbutin are similar cosmetic ingredients which currently not regulated in Regulation (EC) No 1223/2009. Their effects on the skin could be attributed to their gradual hydrolysis and release of hydroquinone (a prohibited ingredient who is only permitted for professional use in artificial nail systems with a maximum concentration up to 0.02%). Respectively in SCCS/1552/15 and SCCS/1550/15, SCCS assessed the safety of alpha- and beta-arbutin. Based on these two opinions and newly submitted data, SCCS concludes in the latest opinion that the use of the two substances in cosmetics is safe as shown below:
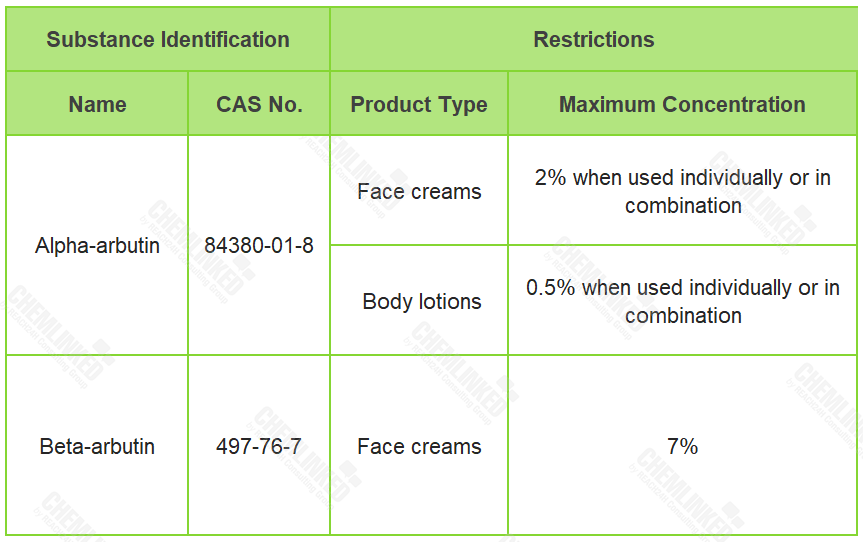 In terms of the hydroquinone contamination requirement, SCCS emphasizes that hydroquinone should remain as low as possible in formulations containing alpha- or beta-arbutin, and should not be higher than the unavoidable traces in both arbutins.
In terms of the hydroquinone contamination requirement, SCCS emphasizes that hydroquinone should remain as low as possible in formulations containing alpha- or beta-arbutin, and should not be higher than the unavoidable traces in both arbutins.



 We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by
We provide full-scale global cosmetic market entry services (including cosmetic registering & filing, regulatory consultation, customized training, market research, branding strategy). Please contact us to discuss how we can help you by 









